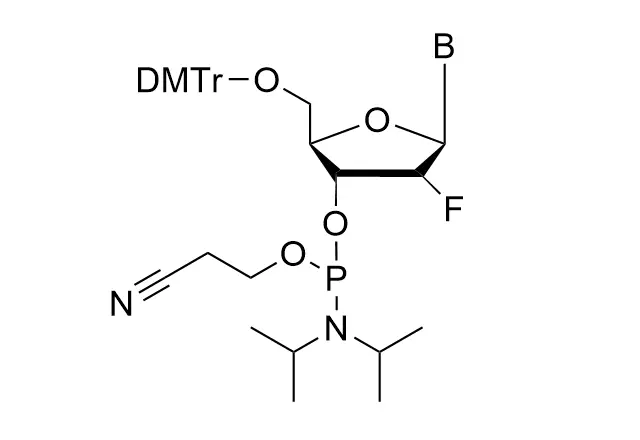
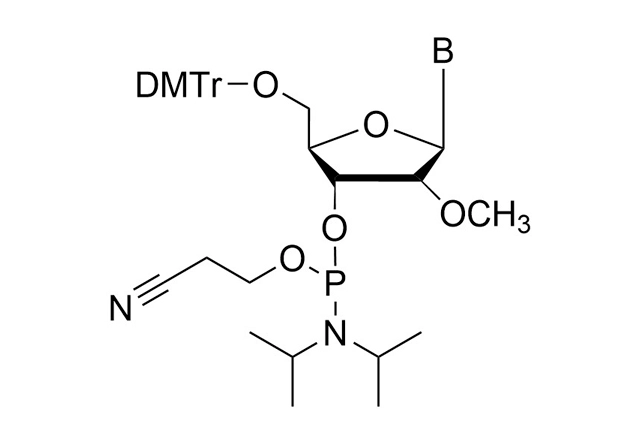
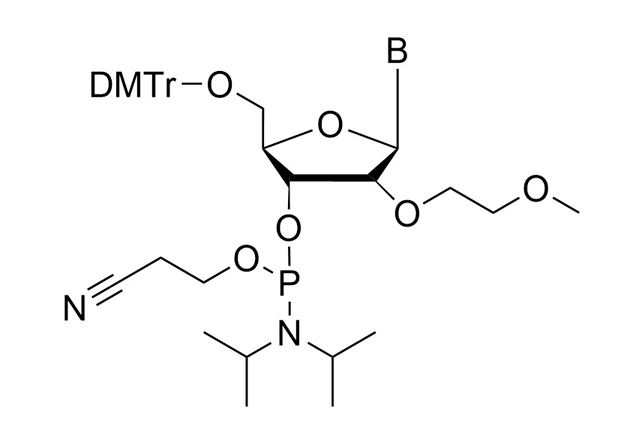
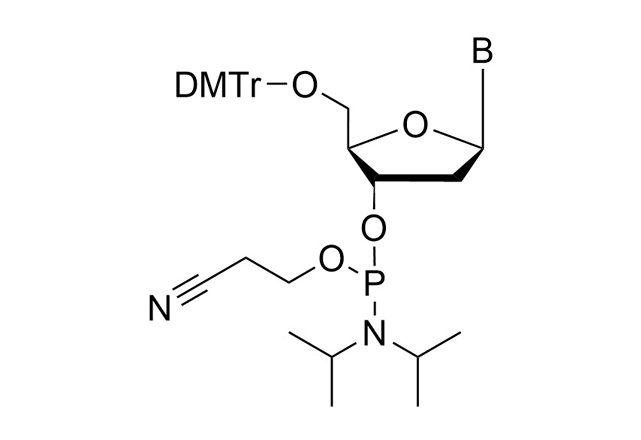
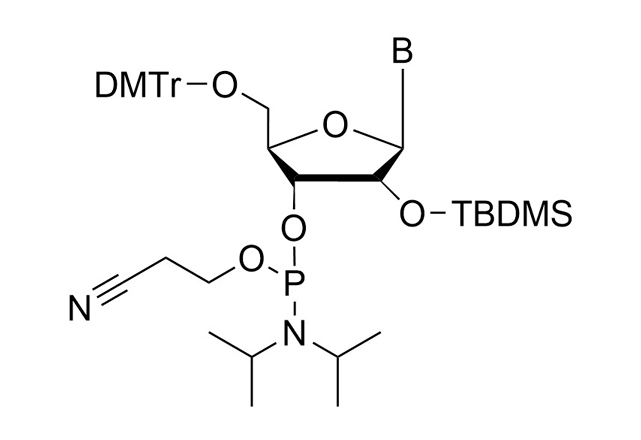
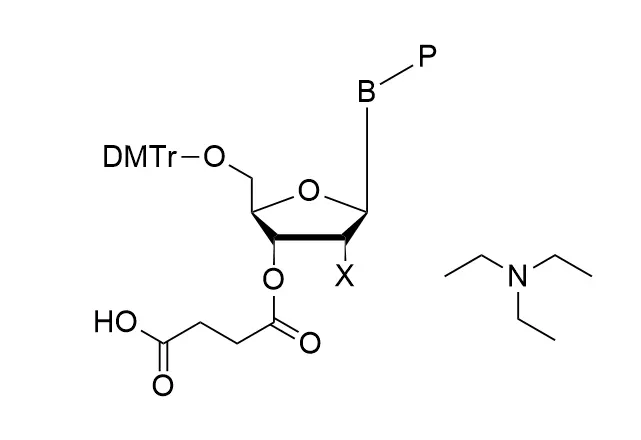
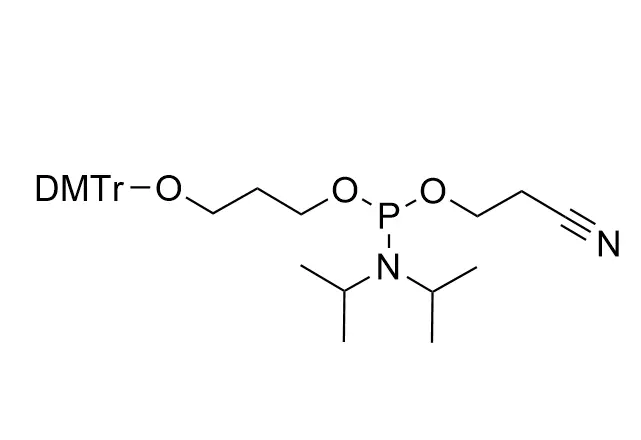
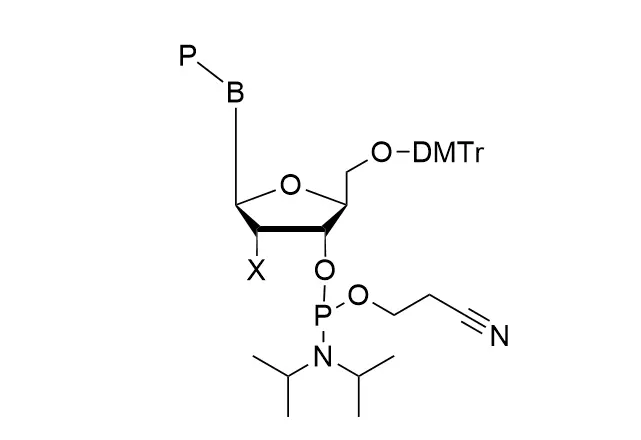
As all of our customers are no doubt aware, phosphoramidites are moisture-sensitive. Indeed, by far the most common cause of poor performance of a phosphoramidite is the presence of moisture.
A critical component of Huaren's quality management system (QMS) is the standard operating procedure (SOP) covering packaging of amidites into vials ready to be attached to synthesizers. When a new batch of a phosphoramidite is produced and approved by QC, the most urgent task is to package it efficiently. The only way to preserve its integrity and maintain a high coupling efficiency even after months of storage, is to package this bulk into end-user vials within a few days. At this point, we use historical sales data along with information on pending orders to determine which pack sizes and vial types to elect to weigh.
We are, of course, able to do special packaging if it is in phase with our production and QC schedules.
Finally a description of our products is mentioned on the Certificate of Analysis that we provide with each product. Specific information about dissolution and special instructions are written on this Certificate, when applicable.So please do not forget to check this important information before using intermediate pharmaceutical products.

 En
En Cn
Cn