Nucleic acid therapeutics are based on nucleic acids or closely related chemical compounds. They include antisence oligonucleotides, aptamers and small interfering RNAs (siRNA), and are typically considered inn cases where specific inhibition of the function of a particular gene involved in disease is thought to be therapeutically desirable. The extrodinary success of mRNA vaccines against coronavirus disease 2019 has renewed interest in mRNA as a means of delivering therapeutic proteinis.
The integration of the recent advances in technologies like mRNA optimization, delivery systems, may unlock the promise of bilogically targeted mRNA therapeutics, beyond vaccines and other immunostimulatory agents, for the treatment of diverse clinical indications.
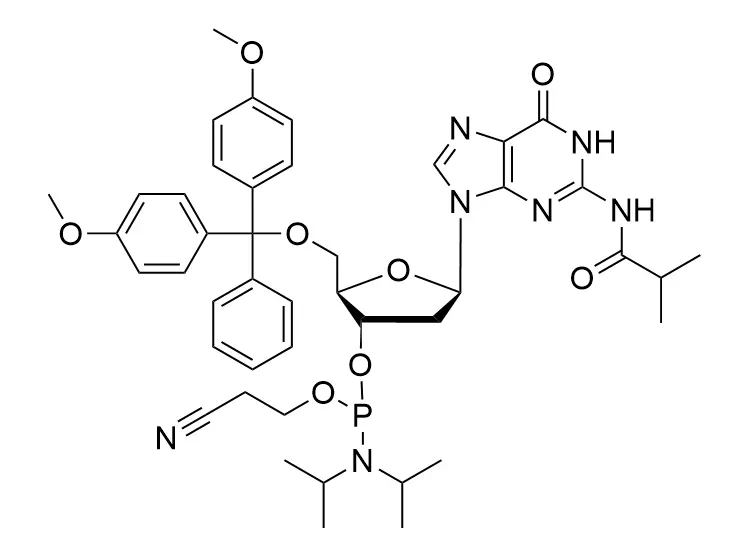
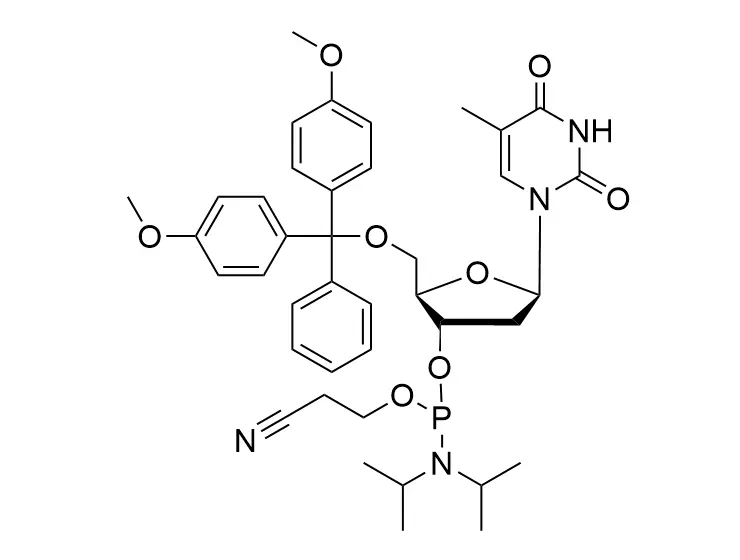
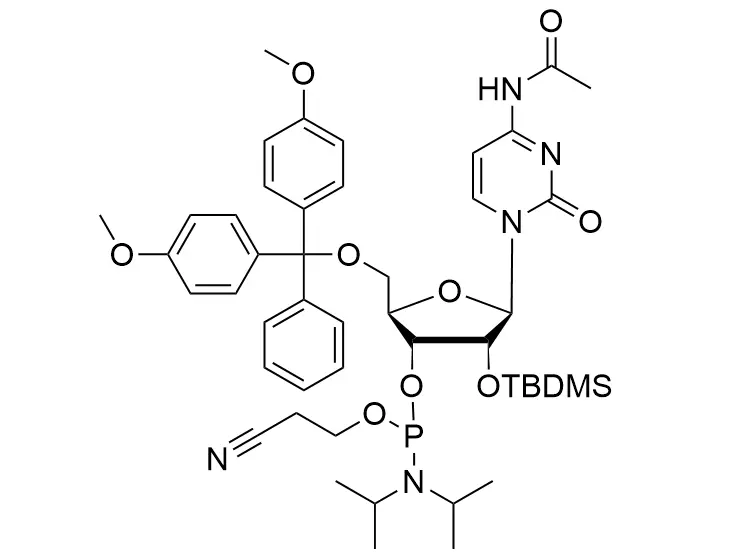
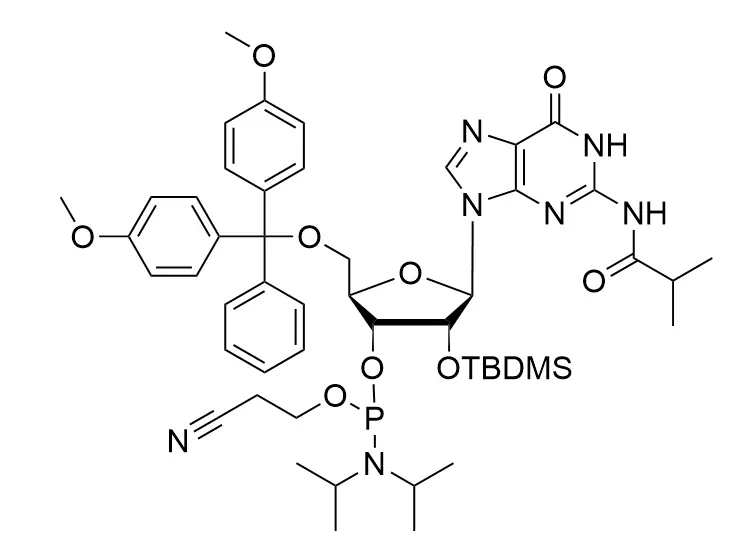
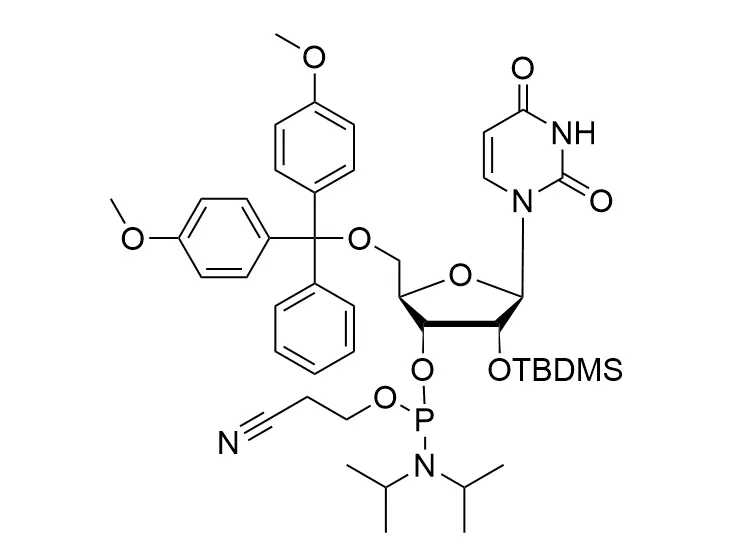
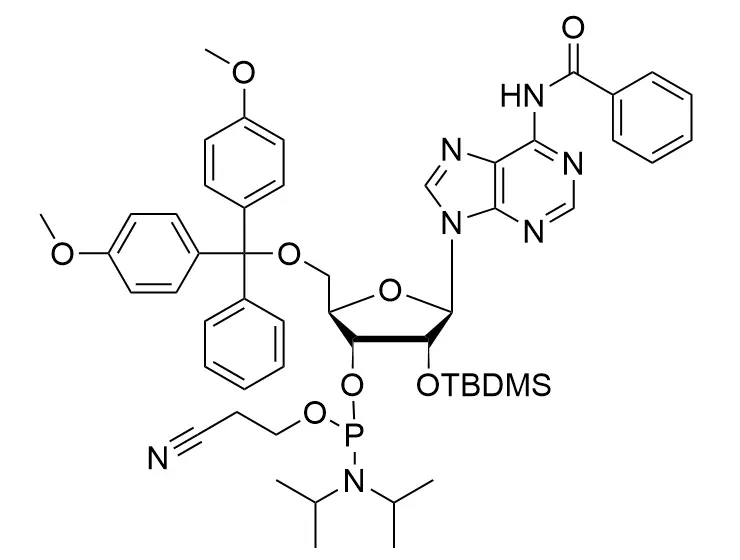
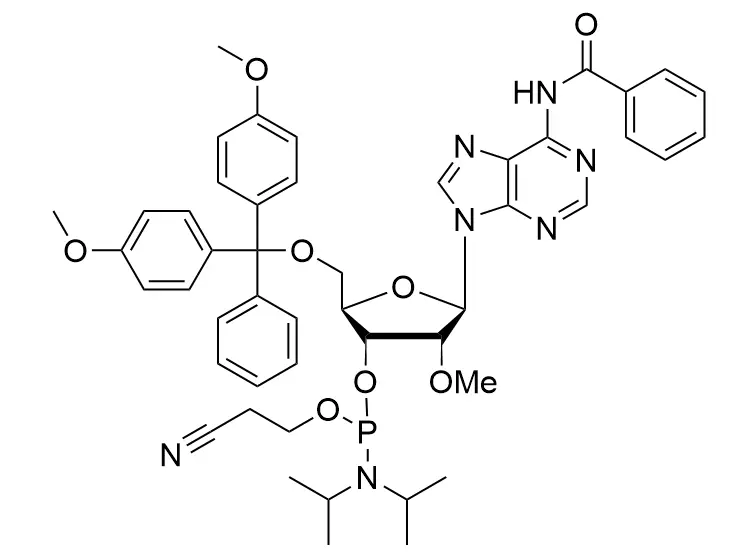
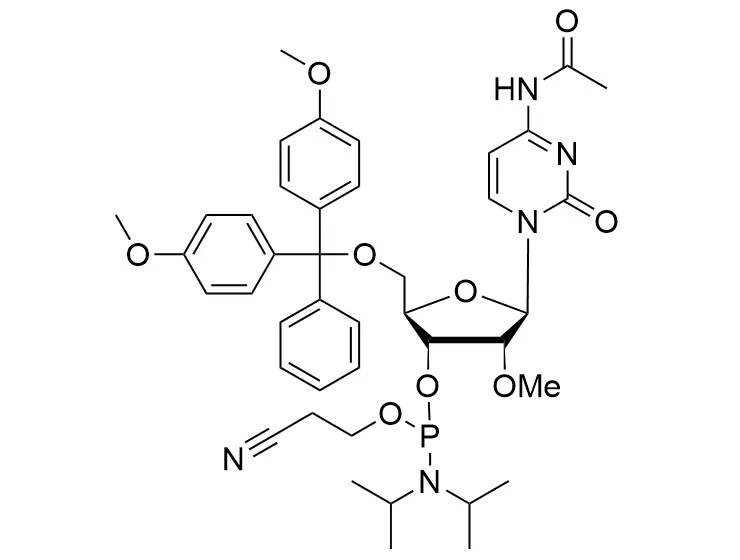
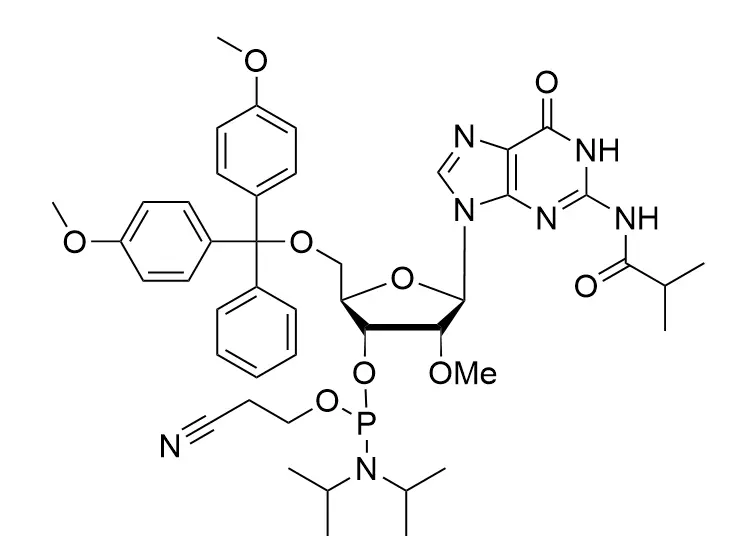
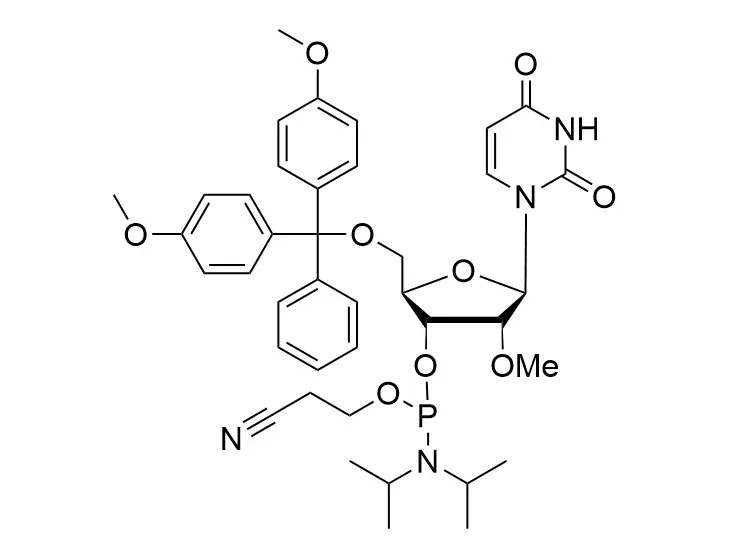
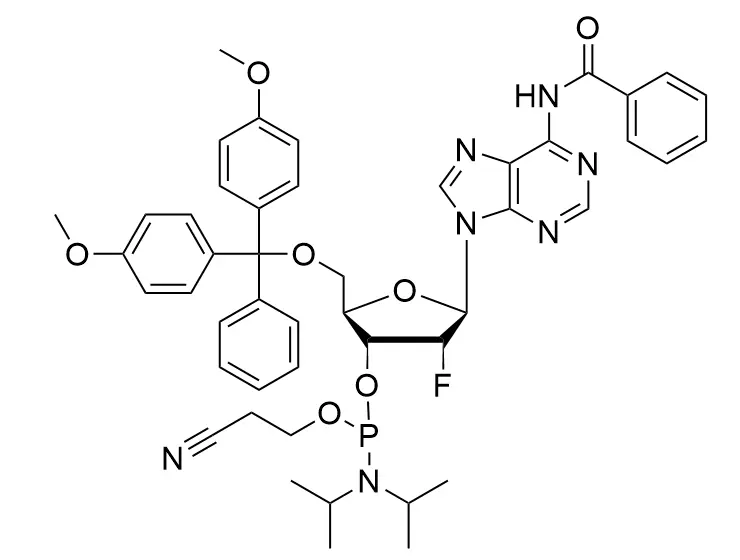
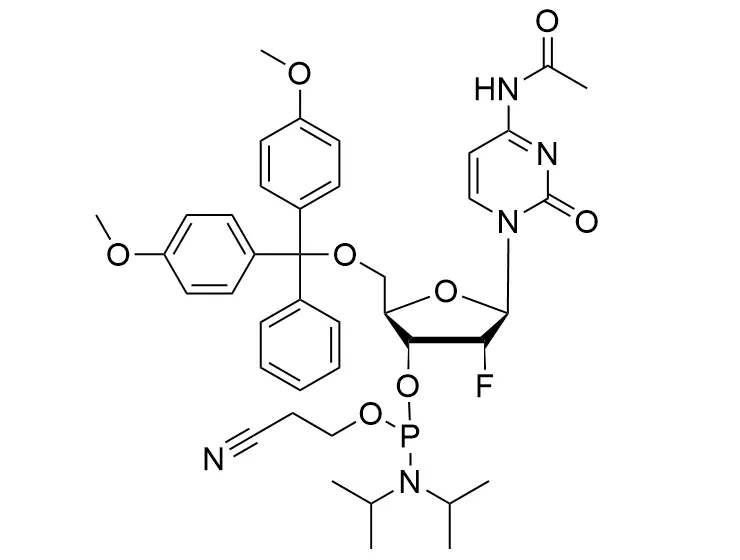
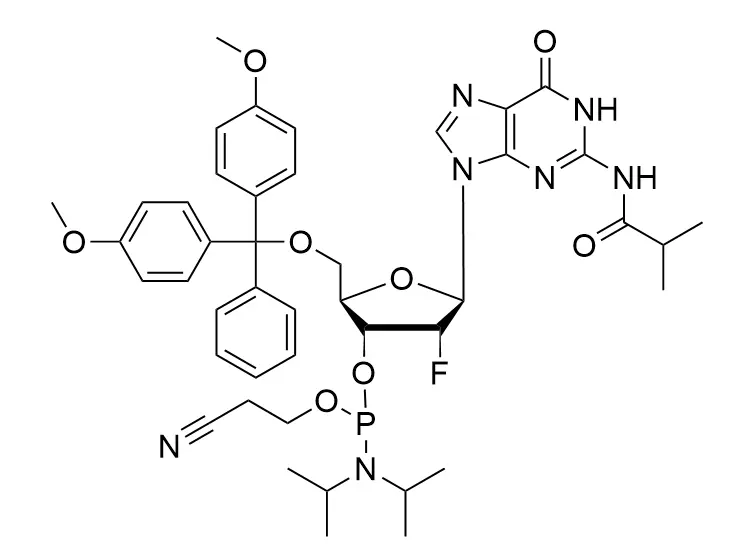
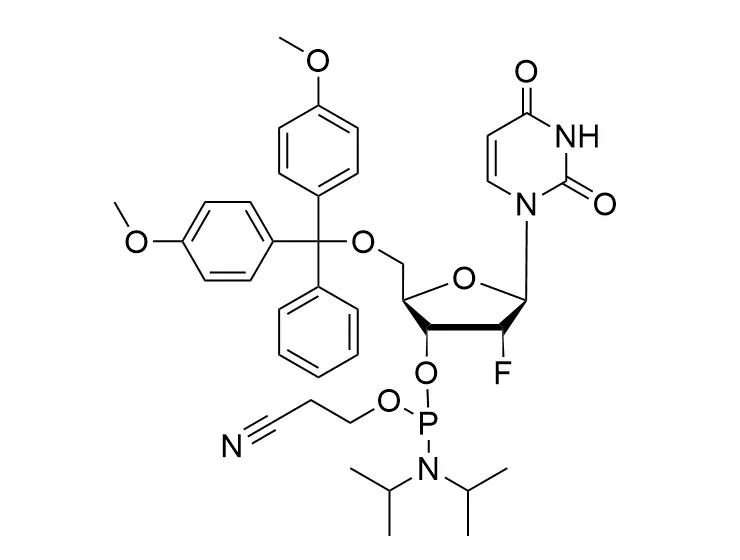
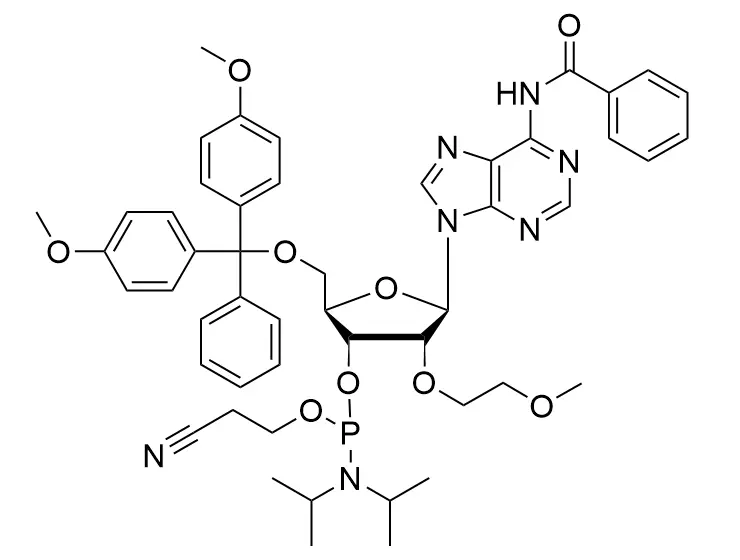
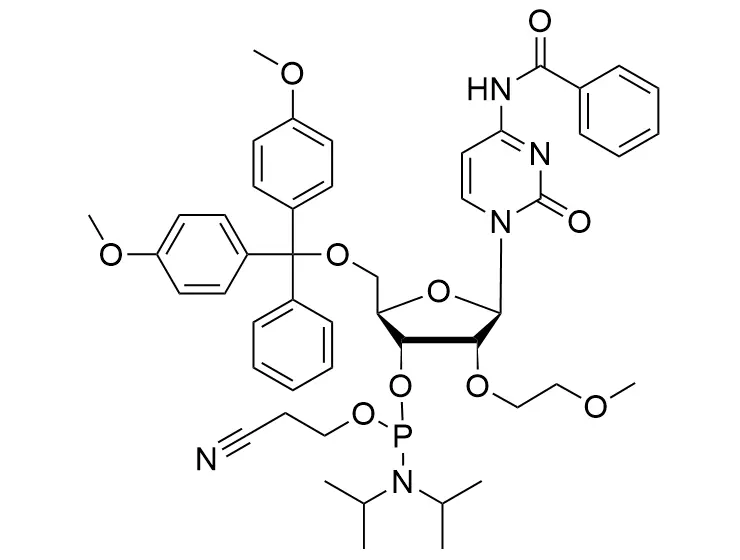
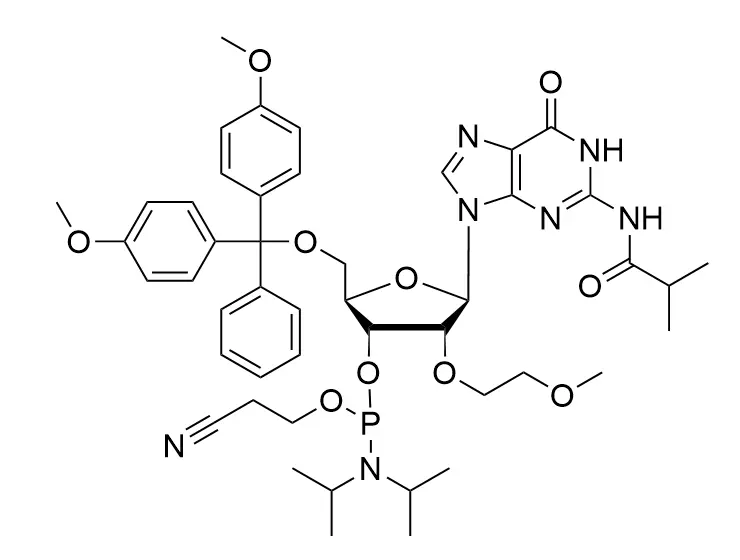
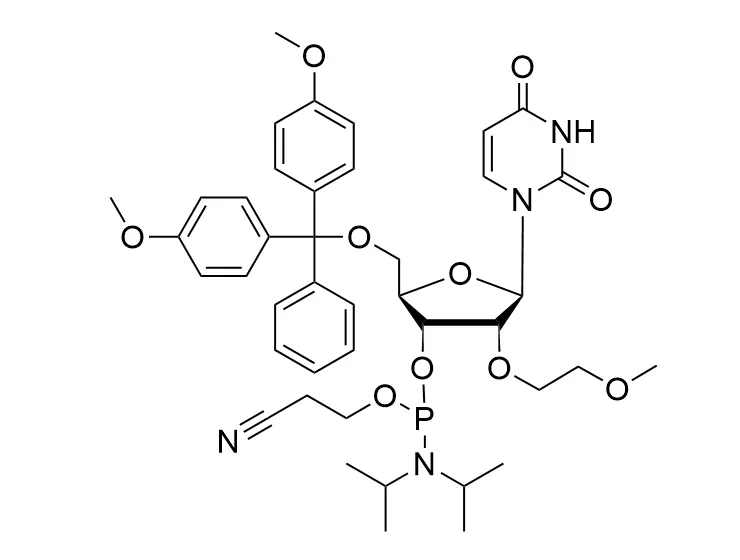
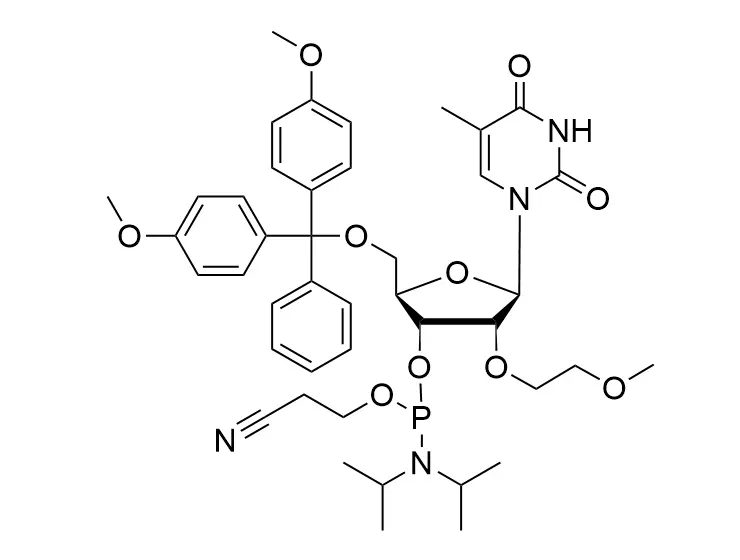
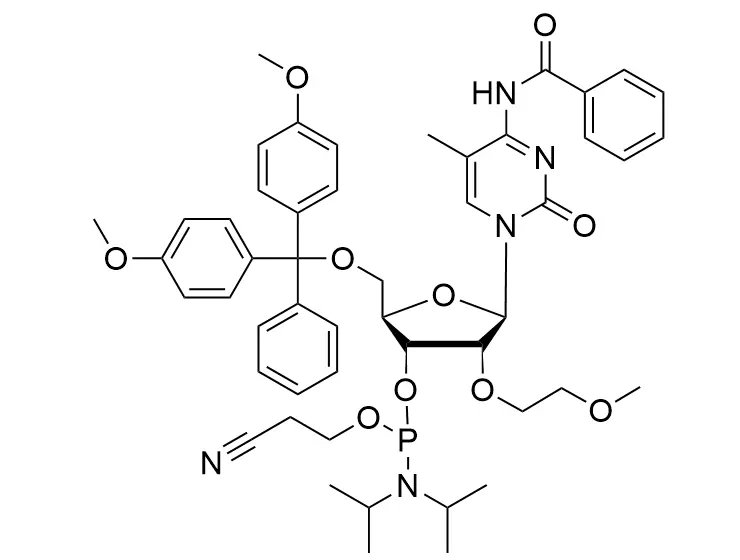
Phosphoramidite chemistry has been the gold standard for oligonucleotides synthesis for decades. Huaren supply oligonucleotides manufacturers and nucleic acid drug R&D companies with RNA phosphoramidites of different protection and modification strategies at high quality and competitive price.
Unmodified DNA and RNA exhibits minimal therapeutic activity because they are rapidly degraded, exhibit poor cellular uptake and/or are filtered out of blood in biological environments. In order to overcome these problems, enormous efforts have been made to identify oligonucleotides chemistries, naturally occurring and synthetic, which improve the target binding affinity, plasma stability, resistance to degradation, and pharmacokinetics. Most of the currently utilized chemistries for oligonucleotides have been resolved and characterized for several years and the fact that they are still widely used implies that they have proven to be highly effective. The structure of an oligonuceltide can be modified at all three of the functional regions—the nucleobase, the carbohydrate, and the phosphodiester linkage.
Nucleobase Modification
By modifying the base of the nucleotide, a higher affinity for the target nucleotide can be achieved. This increases the thermal stability of the duplex formed between the oligonucleotide and its target RNA, which can greatly increase the activity when using oligonucleotides for mRNA-silencing.
Carbohydrate Modification
The deoxyribose in DNA and the ribose in RNA can be modified to increase the oligo's stability against nuclease degradation, greatly enhancing its pharmacokinetic half-life from a matter of days to weeks. This is due to the fact that an electron-withdrawing group on the 2′ carbon of ribose can induce the ribose to pucker into a conformation which is favorable for duplex formation. The most widely utilized modification is the 2'-O-methyl (2'-O-Me) in which a methyl group is attached. Other common modifications include 2'-O-methoxyethyl (2'-O-MOE), and 2'-fluoro RNA (2'-F-RNA).
Phosphodiester Linkage Modifications
The nonmodified phosphodiester (PO) linkage of human DNA and RNA has several unfavorable pharmacokinetic and distribution properties for its use as a therapeutic, including a short half-life in circulation due to nuclease susceptibility and low serum protein binding ability. The most commonly employed chemical modification in both research and clinical use is the phosphorothioate (PS) modification.

 En
En Cn
Cn